Peptide Side Effects: What's Known, What's Unknown, and What People Overstate
A cautious, evidence-aware guide to peptide side effects, long-term unknowns, online claims, and how to read safety discussions without mistaking anecdotes for proof.
Peptide Side Effects: What's Known, What's Unknown, and What People Overstate
The honest answer is that peptide side effects are not one single thing. A side effect profile depends on the specific peptide, the route of exposure, product quality, the person using it, other medications or conditions, and how strong the human evidence is.
That is why broad claims like "peptides are safe" and "peptides are dangerous" are both too lazy. Some peptides are approved medicines with known labeling and post-market safety tracking. Many peptides discussed online are research compounds, compounded products, wellness-clinic offerings, or gray-market products with far less mature safety evidence. Those are not interchangeable categories.
This guide explains how to think about peptide side effects without turning uncertainty into either panic or sales copy. It covers what is commonly reported, what remains unknown, why long-term risk is hard to prove, and how to interpret Reddit threads, clinic claims, vendor pages, and influencer anecdotes.
Educational note: This article is for general information only. It is not medical advice, diagnosis, treatment guidance, dosing guidance, purchasing advice, or a recommendation to use peptides.
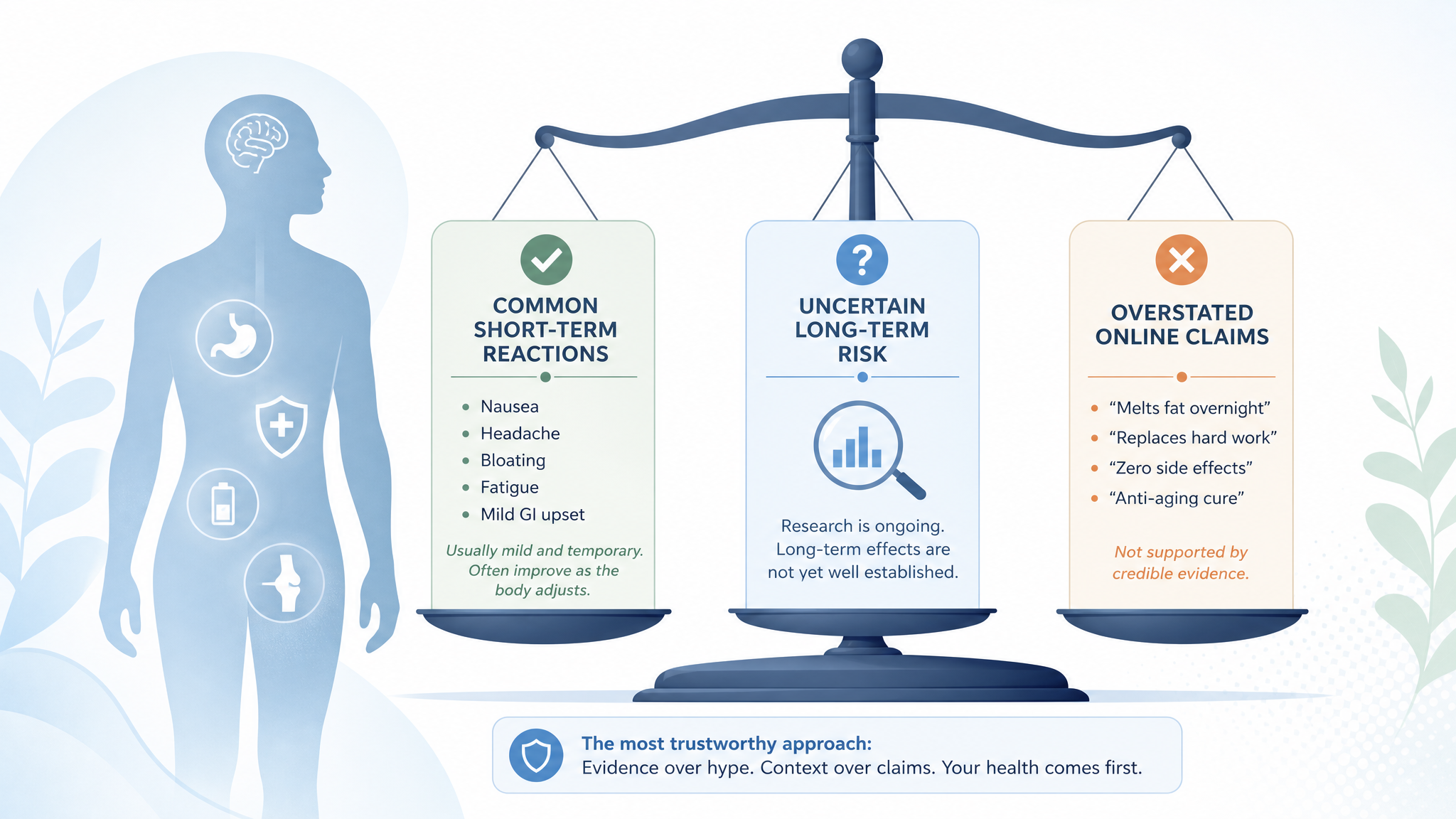 Peptide side-effect claims are easiest to understand when you separate short-term reactions, long-term unknowns, and online claims that run ahead of evidence.
Peptide side-effect claims are easiest to understand when you separate short-term reactions, long-term unknowns, and online claims that run ahead of evidence.
Quick answer: what are common peptide side effects?
The most commonly discussed peptide side effects are usually local reactions, digestive symptoms, fluid or appetite changes, headache, fatigue, sleep changes, and mood-related complaints, but the exact list depends heavily on the peptide and context.
A useful first pass is to separate side effects into five buckets:
- Administration-site problems: redness, soreness, bruising, irritation, infection risk, or tissue injury where an injectable product is used.
- General systemic symptoms: headache, nausea, flushing, fatigue, dizziness, sleep changes, or feeling "off."
- Compound-specific effects: appetite changes, water retention, tingling, changes in libido, skin changes, glucose-related concerns, or hormone-axis effects depending on the peptide class.
- Product-quality risks: contamination, incorrect concentration, degradation, mislabeled ingredients, or sterility concerns.
- Unknown long-term risks: effects that may not show up in short anecdotes, small studies, or early research.
The fifth bucket is the one people skip. "I did not notice side effects" is useful personal information, but it does not prove long-term safety. Absence of obvious symptoms is not the same as mature safety data.
Why peptide side effects are hard to generalize
Peptides are a broad category, not a single drug family with one shared risk profile. A collagen fragment, a GLP-1 receptor agonist, a growth-hormone secretagogue, a wound-healing research peptide, and an immune-signaling peptide can all be called "peptides," but they do not behave the same way.
Three practical differences matter most:
- Target and mechanism. A peptide that affects appetite signaling has a different risk conversation from one discussed for tissue repair or tanning.
- Evidence status. Approved peptide medicines have labeling, trial data, and pharmacovigilance. Research-only or clinic-marketed peptides may have far less human safety evidence.
- Real-world product quality. The safety of a compound on paper does not automatically transfer to an unverified product, an unclear concentration, or a non-sterile preparation.
That is why PeptideBase separates compound pages from broader education. If you are reading about a specific peptide, start with the compound-specific safety page when it exists, such as BPC-157 side effects and unknowns or TB-500 side effects and unknowns. A broad safety pillar can teach the framework, but it cannot replace compound-specific evidence.
What is actually known?
What is known about peptide side effects usually comes from a mix of approved-drug labeling, human trials, small pilot studies, case reports, animal studies, mechanistic research, and real-world anecdote. Those sources are not equal.
A strong safety claim usually needs several things: enough people studied, clear adverse-event reporting, relevant exposure duration, realistic populations, and follow-up long enough to catch delayed problems. Many peptide discussions online do not meet that bar.
For some peptide medicines, side effects are relatively well characterized because they have gone through formal development and monitoring. For many peptides popular in biohacking, recovery, skin, body-composition, or "optimization" circles, the evidence is thinner. That does not automatically mean severe harm is likely. It means the uncertainty is real.
Answer block: known does not mean universal. If a side effect is listed for one peptide class, it should not be copy-pasted onto every peptide. If a side effect has not been reported often, that does not prove it cannot happen. Safety evidence is strongest when it is specific to the peptide, the route, the population, and the exposure pattern being discussed.
What remains unknown about long-term peptide side effects?
Long-term peptide side effects are often unknown because many popular peptides have not been studied in large, diverse human populations over long periods. That is the boring answer, which is usually the correct one.
Long-term uncertainty can include:
- effects from repeated exposure over months or years
- rare adverse events that small studies cannot detect
- interactions with medications or underlying conditions
- effects in pregnancy, fertility contexts, autoimmune disease, cancer history, cardiovascular disease, liver or kidney disease, and complex endocrine conditions
- immune reactions or antibody formation in some peptide contexts
- risks from chronic manipulation of growth, repair, appetite, inflammation, or hormone-related pathways
- cumulative risk from stacking multiple compounds
This is where online certainty gets dumb fast. Someone can say "I used it for eight weeks and felt fine," and that may be true. It still does not answer the long-term question. Eight weeks of personal experience is not a substitute for years of structured safety follow-up.
For more on evidence categories, read Peptide research status explained and what preclinical actually means. Those pages explain why animal findings, mechanism diagrams, and human outcomes should not be treated as the same level of proof.
Why Reddit side-effect threads are useful but limited
Reddit can be useful for finding patterns people are worried about. It is not a controlled safety database.
Anecdotes can help you notice recurring complaints: sleep disruption, mood changes, injection irritation, appetite shifts, bloating, fatigue, or unexpected symptoms people did not see mentioned on vendor pages. That signal is worth reading with curiosity.
But Reddit has major limits:
- people rarely know whether the product was accurately labeled
- people often combine multiple compounds, supplements, medications, and lifestyle changes
- doses, routes, timing, and health context may be missing or unreliable
- people with bad experiences may be more motivated to post
- people with positive experiences may minimize problems because they want the compound to be "worth it"
- replies often confuse confidence with expertise
Answer block: how to read Reddit claims. Treat Reddit as a place to collect questions, not answers. If a side effect appears repeatedly, it may be worth investigating. If one person claims a peptide cured or ruined everything, treat that as a story, not a conclusion.
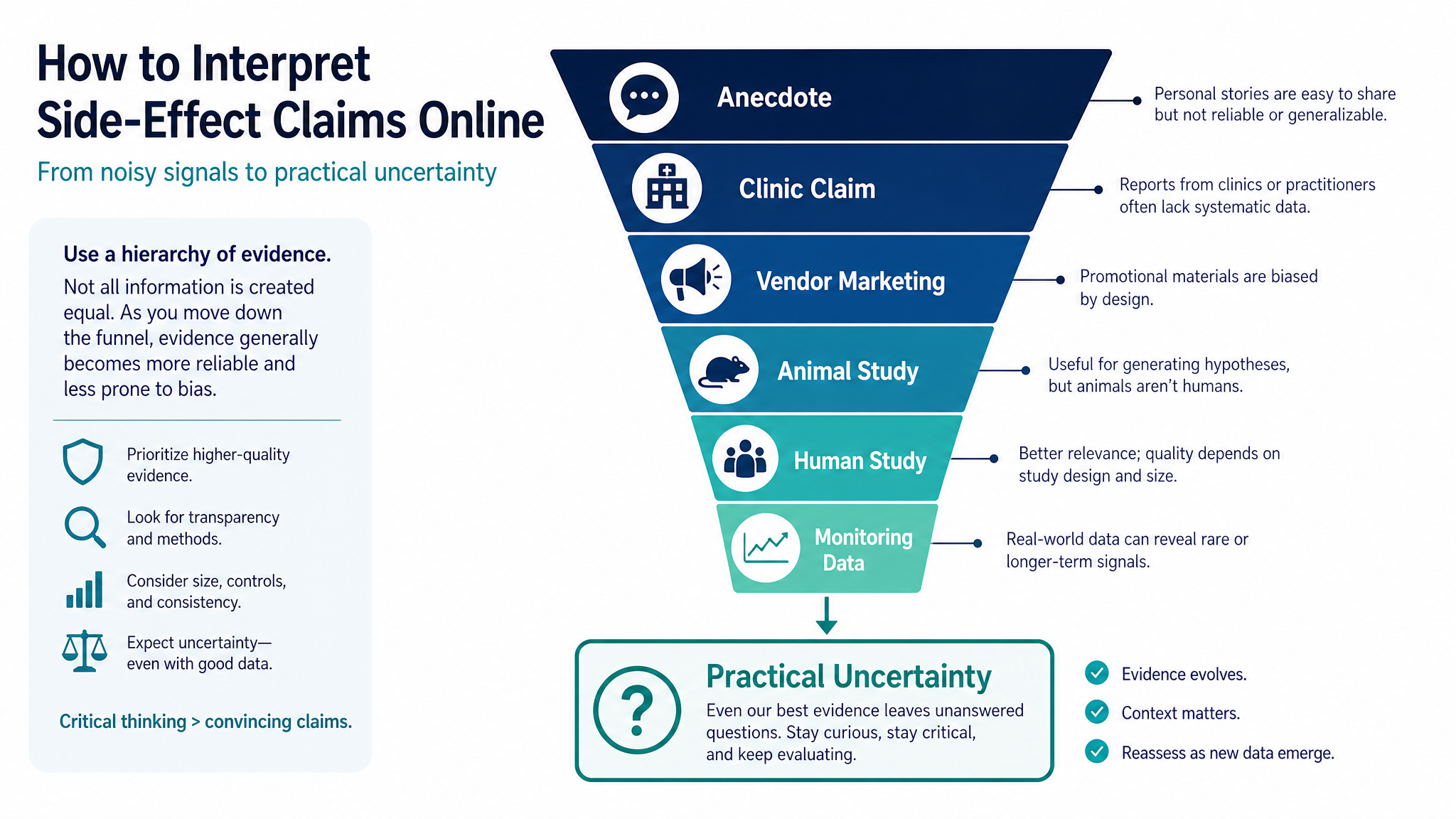 Side-effect claims become more useful when you ask what kind of evidence produced the claim and what context is missing.
Side-effect claims become more useful when you ask what kind of evidence produced the claim and what context is missing.
Clinic and vendor claims can overstate safety in softer language
Peptide marketing does not always sound like marketing. Sometimes it sounds clinical, polished, and calm. That can make weak claims feel safer than they are.
Watch for phrases like:
- "well tolerated" without saying in whom, for how long, or based on what evidence
- "minimal side effects" without naming the source of the safety data
- "naturally occurring" as if natural automatically means risk-free
- "research-backed" when the research is mainly animal or mechanistic
- "physician supervised" used as a substitute for published safety evidence
- "no known serious side effects" when the real issue is limited study size or limited follow-up
The claim may not be completely false. It may just be missing the part that matters.
A better question is: what evidence would have been able to detect the side effect being denied? If the answer is "a few anecdotes, a small pilot study, and a vendor page," the safety claim should stay modest.
For a deeper framework, use how to evaluate peptide claims online. It is one of the most important pages to read before trusting any confident peptide claim.
Side effects in women: what can and cannot be assumed
Searches for peptide side effects in women usually mix real concerns with a shortage of specific evidence. Sex, hormones, body composition, pregnancy potential, menstrual cycle patterns, contraception, fertility treatment, autoimmune risk, and iron or thyroid status can all change how symptoms are interpreted.
That does not mean every peptide has a unique "women's side effect profile." It means broad safety claims should not assume that data from one population answers every question for another.
Practical uncertainty is especially important around pregnancy, breastfeeding, fertility treatment, menstrual-cycle changes, endocrine symptoms, and conditions that are more common in women, such as some autoimmune disorders. Many online peptide discussions do not include enough data to make confident statements for these contexts.
Answer block: peptide side effects in women. The safest educational framing is that women-specific peptide safety depends on the compound, evidence base, reproductive context, endocrine context, and medical history. If a peptide has not been studied well in those settings, confidence should be low, not creatively upgraded.
Mood, sleep, and "feeling weird" complaints deserve careful interpretation
Mood-related peptide side effect claims are difficult because they are real to the person experiencing them but often hard to attribute.
Sleep changes, anxiety, irritability, low mood, restlessness, libido changes, appetite changes, and fatigue can come from many sources: the peptide, expectation effects, illness, calorie changes, training load, other supplements, medication interactions, poor sleep, alcohol, stress, or the simple fact that people start paying closer attention after beginning something new.
The right response is not to dismiss the complaint. The right response is to avoid pretending the cause is obvious.
A cautious way to phrase it is: some people report mood or sleep changes while using certain peptides, but attribution is often uncertain unless the symptom pattern, timing, other exposures, and medical context are carefully assessed. That is less satisfying than a yes/no answer, but it is more honest.
Product quality may be the hidden side-effect variable
Some "peptide side effects" may not come from the peptide itself. They may come from product quality problems.
This is a major reason PeptideBase does not provide sourcing advice. It is also why side-effect reports from gray-market products are so hard to interpret.
Product-quality concerns can include:
- inaccurate labeling
- incorrect concentration
- contamination
- degradation from storage or handling
- non-sterile preparation
- unexpected ingredients
- inconsistent batches
If someone reacts badly to an unverified product, the story may involve the peptide, the product, the route, the preparation, or all of the above. Treating the anecdote as clean evidence about the compound is risky.
What people overstate about peptide side effects
The most common overstatements fall into two opposite camps.
The first camp overstates safety:
- "No side effects."
- "Only amino acids, so it is safe."
- "It is natural."
- "Doctors use it, so there is no issue."
- "Everyone online says it is fine."
The second camp overstates danger:
- "All peptides are research chemicals and therefore reckless."
- "Any side effect means permanent damage."
- "One bad story proves the whole category is unsafe."
- "If long-term evidence is missing, harm is guaranteed."
Both camps skip the hard middle: risk varies by compound and evidence quality, and uncertainty should change confidence, not force a dramatic conclusion.
A practical framework for interpreting peptide side-effect claims
Use this framework when reading any peptide side-effect claim online:
- Which peptide is being discussed? Do not generalize from one compound to the whole category.
- What is the evidence source? Approved label, clinical trial, case report, animal study, mechanistic theory, Reddit thread, clinic blog, or vendor page?
- What population was studied or described? Healthy adults, patients with a condition, animals, cells, athletes, older adults, women, people on other medications?
- What route and product context are involved? Injection, oral, topical, compounded, prescription, research product, unknown source?
- How long was follow-up? Short-term tolerability does not settle long-term safety.
- Are benefits and risks being discussed with the same level of skepticism? If benefits get confident language and risks get vague language, the page is probably selling you a vibe.
- What would change your mind? Stronger human trials, adverse-event reporting, pharmacovigilance, or replicated findings should carry more weight than confident comments.
This is not a treatment checklist. It is a reading checklist. The goal is to avoid being either gullible or reflexively cynical.
FAQ: peptide side effects
Are peptide side effects usually mild?
Sometimes, but "usually mild" is too broad unless you name the peptide and evidence base. Some peptide products have well-characterized side effects. Others have limited human safety data, which makes broad reassurance inappropriate.
Are long-term peptide side effects known?
For many peptides discussed online, long-term side effects are not well established. The absence of long-term evidence should be treated as uncertainty, not proof of safety or proof of danger.
Can peptides affect mood?
Some users report mood, anxiety, sleep, or energy changes while using certain peptides, but attribution is often unclear. Mood symptoms can be influenced by many factors, including other compounds, medications, health conditions, sleep, stress, and expectation effects.
Are peptide side effects different for women?
They can be, but the better answer is that many peptide discussions lack enough women-specific data. Reproductive context, endocrine factors, pregnancy, breastfeeding, autoimmune history, and medication use can all matter. This is a reason for caution, not a reason to invent certainty.
Should Reddit side-effect reports be trusted?
They should be read as anecdotes, not proof. Reddit can reveal patterns and questions worth investigating, but it cannot control for product quality, route, dose, other compounds, medical history, or reporting bias.
Why does PeptideBase avoid dosing and sourcing advice?
Because this site is educational. Dosing, sourcing, and treatment decisions are medical and regulatory issues, and peptide product quality can materially affect risk. PeptideBase focuses on evidence framing, safety uncertainty, and claim evaluation rather than telling readers what to use or where to get it.
Bottom line
Peptide side effects are best understood with disciplined skepticism. Some concerns are common and practical, like local reactions or digestive symptoms. Some are compound-specific. Some are product-quality problems. Some are long-term unknowns that online communities badly underweight.
The safest mental model is simple: specific peptide, specific evidence, specific context. Anything broader than that is probably either marketing, panic, or a forum thread trying to do the job of clinical research.